- THERMOFISHER CASE STUDY
LabVIEW Accelerates Next Generation DNA Sequencing
By Kevin Bodner and Chris Lynch, R&D Engineering, ThermoFisher


OBJECTIVE
Our challenge was to produce a complete software solution to operate a trio of products that prepares samples for next-generation DNA sequencing. The solution needed to include commercial shipping software along with software tools for R&D feasibility, manufacturing and field service. We had to deliver robust software within a compressed schedule of nine months – without an established software development infrastructure.

APPROACH
Leverage the commercial software development tools and expertise from JKI’s NI Certified LabVIEW Architects to create a flexible, extensible suite of software products in LabVIEW. LabVIEW and JKI’s Rapid Application Framework for LabVIEW enabled the seamless development of a fully configurable software solution that satisfied ever evolving hardware and user interface needs.

THE SYSTEM
- NI LabVIEW 2009 SP1
- JKI VI Package Manager Enterprise
- JKI Rapid Application Framework for LabVIEW
- NI PCMCIA-232/4 Serial Interface
- NI USB-6008 and NI USB-6009 Multifunction DAQ
- NI USB-6501 DIO
- NI USB-9162 Module Carrier
- NI 9211 and NI 9213 Thermocouple Input Modules
- NI 9215 AI Module
- NI 9472 DO Module
Background: Sample Preparation for Next Generation DNA Sequencing
There are two basic steps in DNA sequencing: (1) preparing the DNA sample for sequencing, and (2) using a sequencer to detect the DNA sequence of the sample. Typically, sample preparation is a very manual and time consuming process. The The Applied Biosystems™ SOLiD™ EZ Bead™ System for SOLiD DNA sequencing consists of three bench-top instruments: an Emulsifier, an Amplifier, and an Enricher, which automate the otherwise laborious task of preparing a sample for DNA sequencing.
The SOLiD EZ Bead Sample Preparation System
The goal of sample preparation using the SOLiD EZ Bead System for SOLiD sequencing is to prepare the DNA-templated beads that will be deposited onto the glass slide. There are three main steps to doing this, and each is performed by one of the three instruments in the SOLiD EZ Bead System.
Step One – Emulsification: The Emulsifier instrument (Figure 1) consists of a mixer and a peristaltic pump. An emulsion is an oil-aqueous-bead mixture and contains uniformly-sized aqueous droplets, with each droplet containing one bead. The Emulsifier creates the emulsion by metering the rate of aqueous addition and agitating the oil-aqueous-bead mix for a specified time.
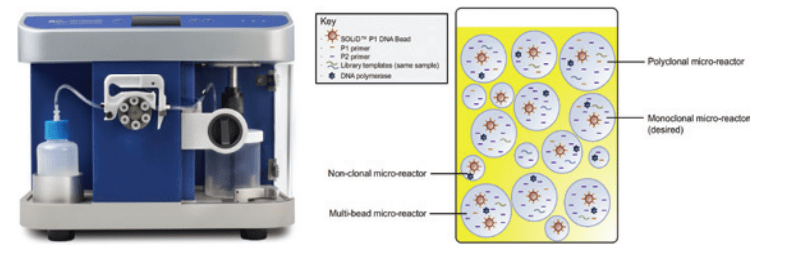
Figure 1: The SOLiD™ EZ Bead™ Emulsifier and a Finished Emulsion of Aqueous Droplets in Oil
Step Two – Amplification: The Amplifier instrument (Figure 2) consists of two Peltier-based, vertically oriented thermal blocks that are sandwiched around a bag of emulsion. Polymerase chain reaction (PCR) chemistry creates many copies of DNA fragments, increasing exponentially, by repeated heating and cooling cycles. The Amplifier performs PCR on the emulsion by ensuring temperatures are ramped rapidly and accurately. The number of cycles used for PCR, along with the temperatures and hold times for each cycle, are easily edited. LabVIEW, running on a netbook computer, controls the Amplifier.
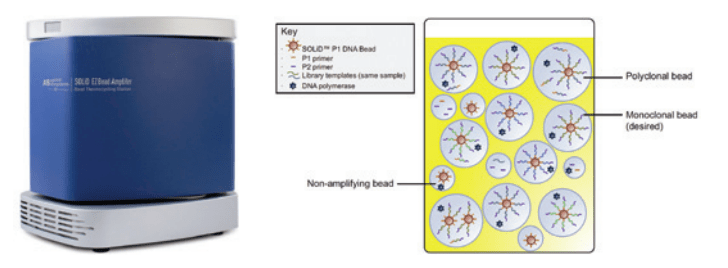
Figure 2: The SOLiD™ EZ Bead™ Amplifier and Post-PCR Emulsion of Amplified DNA Templates
Step Three – Enrichment: The Enricher instrument (Figure 3) consists of a syringe pump, 12-way valves, solenoid valves, vibrator motors, thermomixer, conductivity sensor, door locks and switches, and LED indicators. Enrichment is the extraction of the desirable beads, those that underwent successful amplification, from the non-desirable beads, those that did not amplify. The Enricher separates the amplified emulsion into aqueous and oil phases. The aqueous phase is sent through a filter that captures the successfully amplified beads. Following several washes, the enriched beads are removed from the filter and delivered to a collection vessel. LabVIEW runs on a laptop computer to control the Enricher.

Figure 3: The SOLiD™ EZ Bead™ Enricher and Enriched Template Beads Development of Instrument Control Software for SOLiD™ EZ Bead™ System
LabVIEW has played an important role during the last ten years at The Applied Biosystems™, used to develop software during early instrument prototyping and then for manufacturing test and service. The SOLiD EZ Bead software was the first of its kind, as LabVIEW was used to develop the application used by external customers. The use of LabVIEW and VIPM for both the prototype and customer software enabled code reuse while eliminating the need to rewrite software in another programming language. This code reuse allowed us to shorten our development cycles and meet our aggressive schedule.
There were many hardware configurations during the development of the instrument that needed to be supported by the software. Initial breadboards and prototypes made use of off-the-shelf hardware from NI. Direct serial communication to several components was done through a NI PCMCIA-232/4. Digital inputs and outputs were provided by NI USB-6008 Multifunction IO devices. Temperature monitoring was done using NI 9211 and NI 9213 Thermocouple Input Modules housed in NI USB-9162 Module Carriers. For the shipping instruments, a custom electronics board and embedded firmware were used, with a single USB connection. The numerous hardware permutations presented a constant challenge for the software.

LabVIEW and JKI’s RAFL architecture enabled a small software team to deliver commercial software within a nine month period.
Kevin Bodner - R&D Engineering, ThermoFisher
R&D scientists and engineers demanded that the software worked continuously on a variety of different hardware configurations, and remained online 24x7 over time as the hardware evolved. We selected JKI’s Rapid Application Framework for LabVIEW (RAFL) to meet this challenge. It contains a rich set of built-in features to quickly develop professional and robust commercial software. RAFL has built-in support for parallel processes, scripting, multiple user screens, and asynchronous communication and control.
RAFL utilizes LabVIEW object-oriented programming to build extensible classes of subsystems. Hardware classes were defined for generic hardware components, such as a thermal mixer. Then, as the thermal mixer hardware changed, the methods of the generic thermal mixer could be dynamically dispatched to the method for the new thermal mixer (Figure 4).
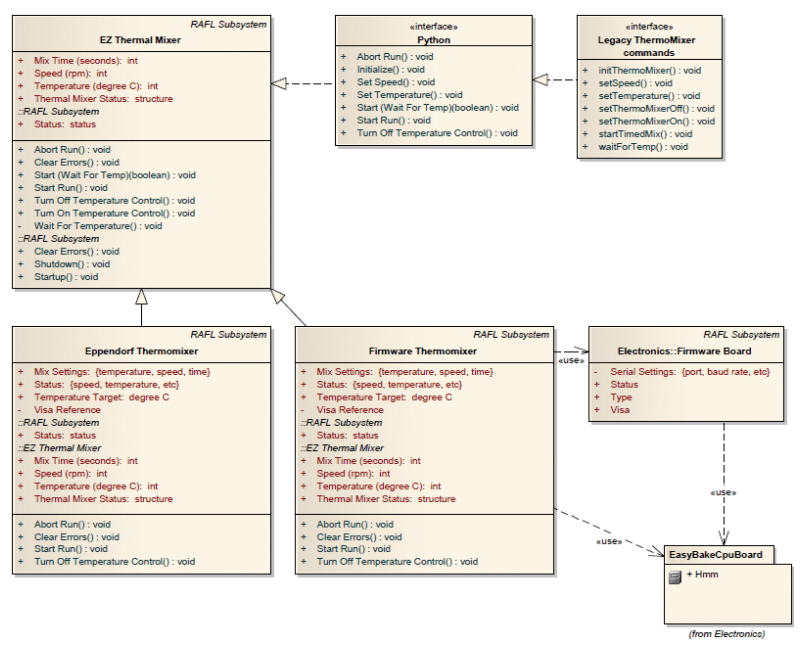
Figure 4: UML Sketch of a Generic RAFL Subsystem and Specific Hardware Implementations
The development of all three modules needed to be completed on a compressed schedule with limited resources. As a result, we resorted to a pragmatic approach to software development. We employed best in class software engineering practices and tools as time allowed, based on informal risk assessments.
We used several Agile practices, scrums to facilitate communication and short sprints to prioritize features and break the work down into smaller tasks. Unified Modeling Language (UML) sketches were used during design, while JKI engineers created an invaluable automated software unit test and build tool. Code reuse was enabled by using JKI’s VI Package Manager (VIPM) to create and manage packages of software libraries. The software team tracked defects during development, while a single software test resource created test plans and executed test cases.
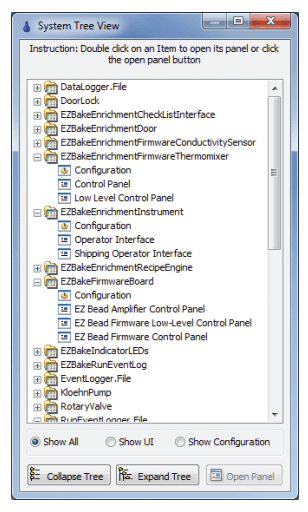
Figure 5: The RAFL System View Allows Access to Low Level Screens
Research and Development Software
R&D software needs to expose all available functions of the hardware to the user. The user requires this level of control to determine the optimal operation of the hardware. RAFL’s built-in system view (Figure 5) provided ready-access to low level manual control and configuration screens for each subsystem. This mechanism enabled users to manually control each subsystem individually and to troubleshoot any problems with connections, communications, or configurations.
After the optimal operation of subsystems was determined, they were combined to create chemistry protocols. Scripting was key to enabling the efficient development of chemistry protocols. RAFL has built-in support for both Python™ and Standard Commands for Programmable Instruments (SCPI) through Transmission Control Protocol (TCP) connections. Python, an established programming language, was chosen for its simplicity while enabling conditional statements, looping, and passing of variable parameters. Scientists developed custom Python scripts for all protocols. Python scripts send commands to a server written in LabVIEW, listening on a TCP port, which in turn directs commands to the appropriate subsystem.
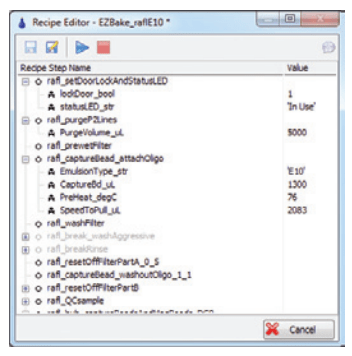
Figure 6: R&D Recipe Editor
Once the basic scripts were finished, the R&D Recipe Editor Screen (Figure 6) could be used to modify the parameters and run the script. In this case, LabVIEW loaded and edited Python scripts and sent them to Python for execution.
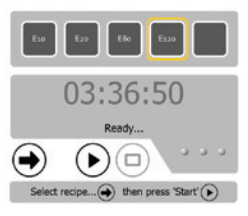
Figure 7: SOLiD™ EZ Bead™ Amplifier Running on Netbook
Customer Software
A simplified user screen (Figure 7) and workflow were needed for the shipping customer software. Also, the look and feel of the screens needed to be consistent (Figure 8) across all three modules. Again, flexibility was a key contributor to success. We knew final hardware and firmware was not going to be ready in time for release to early access customers, but RAFL helped us work around this issue. The early access customers used the final customer screen, without the final instrument configuration. This strategy allowed us to collect important feedback on both the software and instrument. Instrument configurations are defined in Extensible Markup Language (XML) files and the required XML file is loaded at startup. As a result, the same customer screen can be used to control any combination of hardware components.
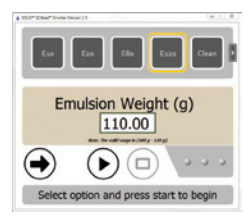
Figure 8: SOLiD™ EZ Bead™ Enricher Running on Laptop
Manufacturing Test and Service Software
The manual control screens for all of the subsystems enabled manufacturing test and servicing of the instrument. When a need surfaced to test the Enricher fluidic system as a whole, a new screen (Figure 9) was developed. As in R&D, custom Python scripts were created for manufacturing tests.
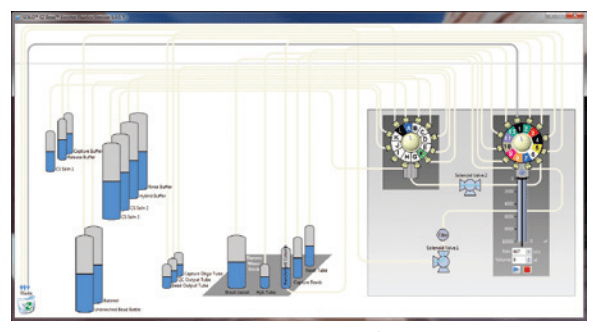
Figure 9: Fluidics Test Screen
Benefits Realized
With the release of the EZ Bead system, SOLiD customers can realize an 80% reduction in sample preparation time, with less than 1 hour of hands-on time now required. Over 200 SOLiD EZ Bead Systems have been shipped. Combined, customers are performing over 1000 runs per month. Six months after product launch, no software patches or upgrades have been required.

LabVIEW and JKI’s Rapid Application Framework for LabVIEW enabled the seamless development of a fully configurable software solution that satisfied ever evolving hardware and user interface needs.
Chris Lynch - R&D Engineering, ThermoFisher
LabVIEW and JKI’s Rapid Application Framework for LabVIEW architecture enabled a small software team to deliver commercial software within a nine month period. This achievement resulted in a reduced software development expenditure that will result in a shorter payback period and an increased return on investment. Further, the flexibility and scripting capabilities of the software allows continued protocol development, without support from R&D software engineers.

5791 Van Allen Way
Carlsbad, CA, 92008
760.603.7200
thermofisher.com

JKI is a National Instruments Certified Silver Alliance Partner.
